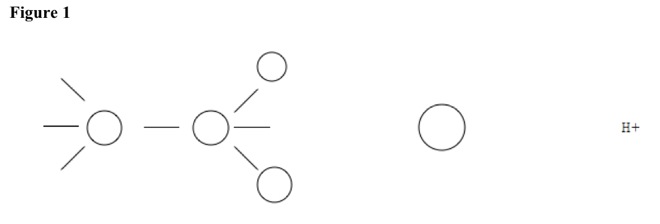
Organic reactions {substitution reaction, organic} can substitute nucleophiles. In reactant, two carbons share one bond, and secondary or tertiary carbon has small and weak nucleophile. See Figure 1.
substitution type 1
Bigger and stronger nucleophile can substitute for weaker and smaller nucleophile, at rate that depends on reactant concentration {substitution type 1} (SN1).
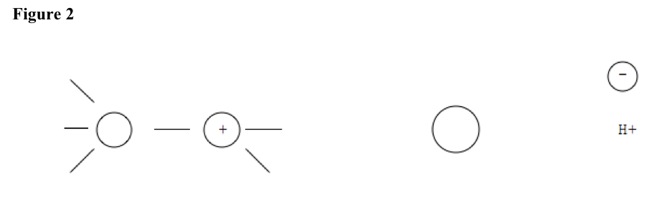
In slow step, polar solvent separates substituent from molecule, making secondary or tertiary carbocation. Secondary or tertiary carbons have more polarity, have larger substituents, and prevent pushing more, compared to primary carbons. See Figure 2.
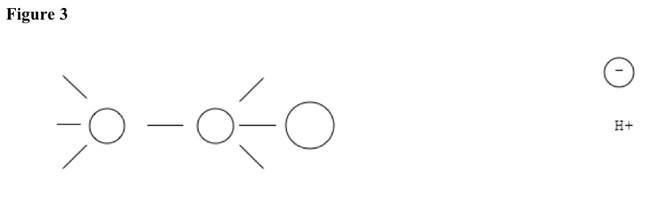
In fast step, other substituent substitutes for separated substituent, helped by polar solvent. See Figure 3. SN1 reactions are not stereospecific, because pull can be from any side.
For example, hydroxyl or cyanide substitutes for halide. Amine substitutes for hydroxyl.
substitution type 2
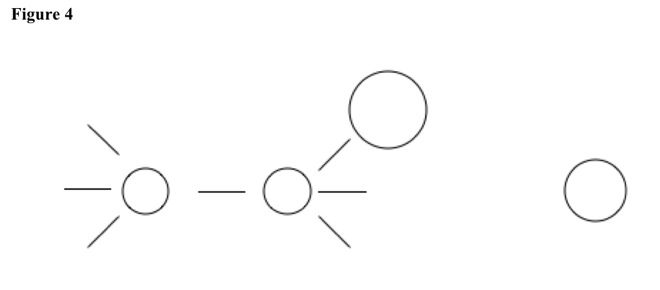
In reactant, two carbons share one bond, and primary carbon has large and strong nucleophile. See Figure 4.
Weak and small nucleophile can substitute for bigger and stronger nucleophile, with rate that depends on two reactant concentrations {substitution type 2} (SN2).
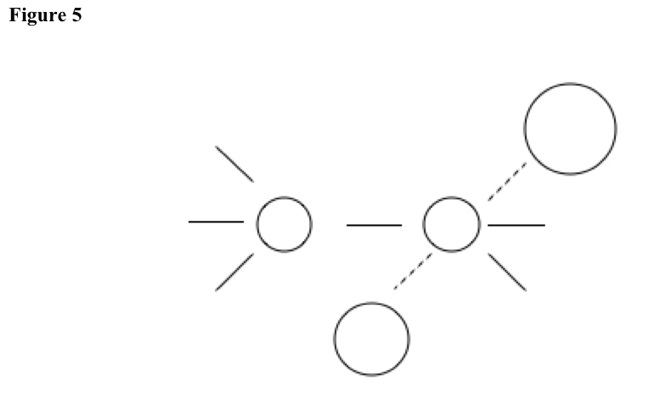
First, electric repulsions from second molecule push away substituent from first molecule, giving intermediate state. Intermediate state is planar, because five substituents attach to carbon. See Figure 5.
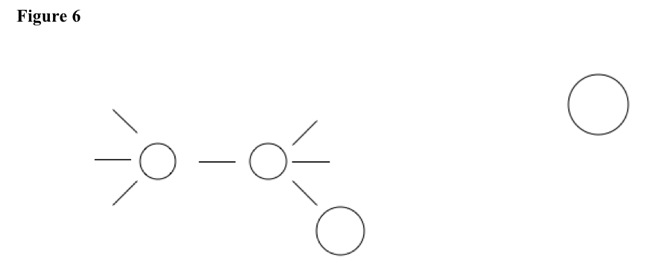
Old substituent leaves, helped by non-polar solvent. SN2 reactions have stereospecific product, because pushing can be from one side only. See Figure 6.






Physical Sciences>Chemistry>Organic>Chemical Reaction
5-Chemistry-Organic-Chemical Reaction
Outline of Knowledge Database Home Page
Description of Outline of Knowledge Database
Date Modified: 2022.0224